Blood Cards
Data Sheet
![]() Shop this product in our online store
Shop this product in our online store
Products - Protein Microarrays - Blood Cards and Blood Card Kits for Collecting, Stabilizing, Sampling and Storing Whole Human Blood Samples for Protein Microarray Assays

Arrayit whole human blood collection cards facilitate the collection, sampling and storage of whole blood and serum components for protein microarrays, immunoassays, allergy testing and other downstream applications that require high quality whole blood samples. Cards contain 4 x 12.5 mm circles on a proprietary collection matrix to enable the collection of 4 x 2-3 drops (50 µl) of whole blood in each of the 4 collection regions. Buffers, columns, lancets, and collection kits simplify the rapid and affordable deployment of Arrayit whole blood cards for a wide range of life sciences, research, veterinary medicine and pathology applications. Whole blood cards and kits are particularly especially useful in protein microarray and enzyme-linked immunosorbent assay (ELISA) applications that have traditionally used purified serum as the sample type. Private labeling is available for large accounts.
Table of Contents
- Introduction
- Quality Control
- Product Description
- Product Contents
- Whole Blood Contents
- Product Protocols
- Technical Assistance
- Troubleshooting Tips
- Recommended Products
- Ordering Information
- Warranty
Introduction
Arrayit has developed an advanced Whole Blood Collection Card for diverse downstream laboratory applications, most notably protein microarrays. These whole blood collection cards can be used to acquire and stabilize RNA, DNA and protein from lancet "finger-stick" collections. The best and most sensitive assays require sample input of the highest integrity. The most important aspect of any dry blood spot collection card is the purity of the collection matrix that the blood sample is applied. There matrix must be free of contamination that will compromise the collected sample. The materials used to manufacture the collection matrix employ the highest purity deionized water and solvents available, and all are validated by chromatography. The final collection matrix holding the whole blood is free of naturally occurring RNases, DNases, and proteases, which are the enzymes that degrade these important biomolecules. Arrayit whole blood cards are assembled in class 1 clean rooms for the most exacting applications. Collection matrices are homogenous in composition, and display uniform thickness and absorbency characteristics across all cards.
Quality Control
Arrayit whole blood cards are manufactured using the highest level of quality control (QC) and quality assurance (QA) available. Quality control and quality assurance of each manufactured lot ensures that our whole blood collection cards and kits perform at the highest levels.
Product Description
Arrayit whole blood card technology offers an advanced card-based whole blood collection method that collects and dry stabilizes whole blood components for a wide variety of downstream applications including protein microarrays. Users will appreciate the following features of this product:
- Manufactured using chromatography-validated raw materials in an ISO9001 environment.
- Collection, drying, stabilization, storage and ship-ready in a single product
- Compatible kit takes guesswork out of sample preparation for protein microarrays
- Low background and excellent signal strength in microarray immunoassays
- Robust whole blood collection capacity
- Compatible with all types of whole blood samples and collection methods
- Bar coded for precise identification
- Compact size
- Covered design protects collected sample from environmental damage.
- Collection instructions provided on every card.
- Manufactured and packaged in class 1 cleanrooms
- Free of biological and chemical contaminants
- Reduces sample shipping costs and conserves laboratory space
Whole Blood Card Collection Kit (Cat. DBCK) Contents
- 100 x Whole Blood Collection Cards
- 100 x Spins Columns
- 100 x 1.5 ml Collection Tubes
- 100 x Lancets
- 1 x 50 ml 1X Elution Buffer
- 1 x 50 ml 1X Reaction Buffer
- 1 x Disc Punch (12.5 mm)
Whole Human Blood Contents
- 50 µl of whole human blood
- Fully saturated 12.5 mm dotted circle
- 2.5 drops whole human blood
- 2.4 x 10^8 red blood cells
- 3.7 x 10^5 white blood cells
- 25 µl of serum
- 1.8 mg total serum protein
- 800 µg total serum protein
- 8 µg white blood cell total RNA
- 1.5 µg total genomic DNA
Short Protocol (Steps 1-10)
- Stick finger tip with lancet.
- Saturate each 12.5 mm dotted circle with 2.5 drops whole blood.
- Allow the card to dry 60 min.
- Excise whole blood card circle using a 12.5 mm disc punch.
- Wet 12.5 mm disc with 60 µl 1X Elution Buffer.
- Place wetted 12.5 mm disc in Spin Column.
- Place Spin Column in 1.5 ml Collection Tube containing 60 µl 1X Reaction Buffer.
- Spin 3 min to collect whole blood sample and mix by vortexing.
- React whole blood sample with protein microarray for 60 min.
- Process and scan the protein microarray to obtain test results.
Complete Protocol (Steps 1-10)
1. Stick finger tip with lancet. Wash your hands for 30 sec with soap and warm water. Rinse hands thoroughly for 30 sec with warm water and dry completely with a lint-free laboratory wipe. Decontaminate the laboratory bench by wiping with 100% ethanol. Place the lancet on the decontaminated laboratory bench and rest the tip of your left hand middle finger gently on the top of the lancet. Press down firmly on the lancet to pierce your middle finger tip.
2. Saturate each 12.5 mm dotted circle with 2.5 drops whole blood. Massage whole blood droplets out of your finger tip using your right hand to apply firm pressure to your left middle finger. Repeated firm pressure strokes from the base to the tip of your finger will accelerate blood flow. Saturate each 12.5 mm dotted circle completely such that the blood migrates outward and slightly beyond each dotted circle. Typically 2-3 drops of whole blood placed in the center of the dotted circle are sufficient to saturate each 12.5 mm collection area. A slight excess of whole blood is preferable because the 12.5 mm disc punch excises precise 12.5 mm disc regions, allowing well-calibrated sample collection volumes if each 12.5 mm dotted circle is wetted completely. If whole blood is being applied to each dotted circle with a pipette, use 50 µl whole blood per 12.5 mm collection region. A properly collected card is shown in Figure 1.
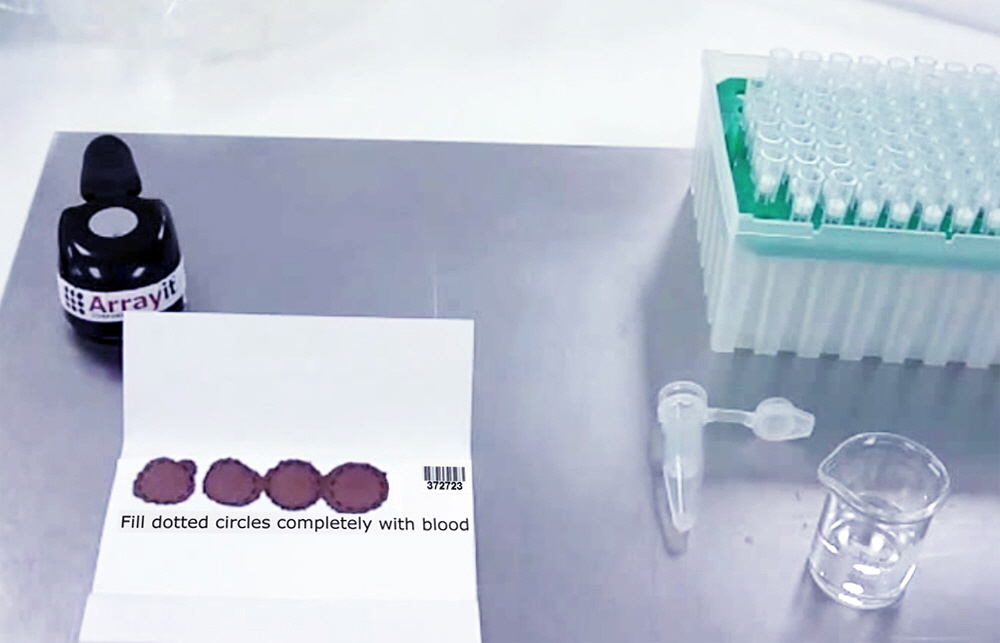
Figure 1. Arrayit Whole Blood Card with properly collected whole blood filling each of the four 12.5 mm dotted circle collection regions. Each collection region has a capacity of approximately 2-3 drops (50 µl) of whole blood.
3. Allow the card to dry 60 min. After filling each of the four 12.5 mm dotted circle collection regions with whole blood, fold the top flap of the card inside the bottom flap and allow the collected blood spots to dry for 60 min at room temperature. The drying process removes water from the collected samples and stabilizes the collected samples. After the 60 min drying period, progress immediately to step 4. Alternatively, the cards can be placed inside a sealed plastic bag and stored at room temperature, 4°C, -20°C or -80°C for future use.
4. Excise whole blood card circle using a 12.5 mm disc punch. The dried whole blood spots can be excised using a 12.5 mm disc punch. Align the punch with the dotted circle at each collection location and press the disc punch firmly to excise the collection matrix containing the whole blood sample. Many microarray assays require only a single 12.5 mm disc, which allows the remaining three discs to be saved and stored for future use. The disc punch should be washed with water and dried with a lint-free laboratory wipe between each card to prevent sample cross-contamination.
5. Wet 12.5 mm disc with 60 µl 1X Elution Buffer. After the 12.5 mm disc containing the whole blood sample is excised with the disc punch, pick up the disc with fine-tipped forceps and wet each side of the 12.5 mm disc with 30 µl of 1X Elution Buffer (60 µl total). To simplify the pipetting process, a single 60 µl volume of 1X Elution Buffer can be loaded into the pipette tip and dispensed onto the two sides of the 12.5 mm disc as two approximately equal 30 µl volumes as shown in Figure 2.

Figure 2. Arrayit Whole Blood Card 12.5 mm collection disc excised and being wetted with 60 µl of 1X Elution Buffer.
6. Place wetted 12.5 mm disc in Spin Column. After the 12.5 mm collection disc is wetted with 60 µl of 1X Elution Buffer, place the disc into a Spin Column using fine-tipped forceps to press the wetted disc into the Spin Column orifice as shown in Figure 3.
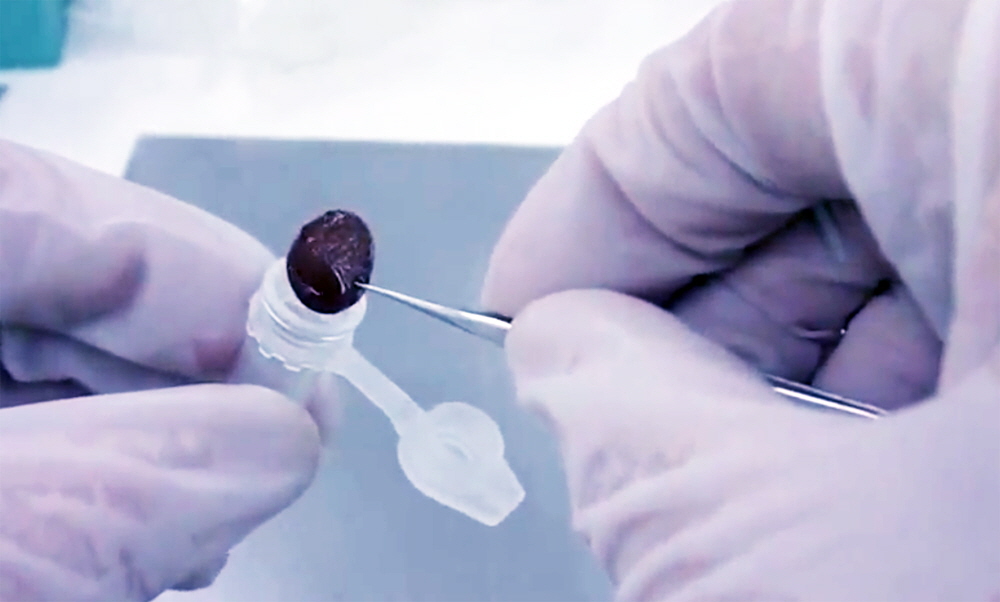
Figure 3. Arrayit Whole Blood Card 12.5 mm collection disc containing 60 µl of 1X Elution Buffer being placed into a Spin Column. The Spin Column is placed inside a 1.5 ml Collection Tube as described in Figure 4.
7. Place Spin Column in 1.5 ml Collection Tube containing 60 µl 1X Reaction Buffer. Pipette 60 µl of 1X Reaction Buffer into the bottom of a 1.5 ml Collection Tube and place the Spin Column containing the 12.5 mm collection disc into the 1.5 ml Collection Tube to obtain the assembly shown in Figure 4. Close the Spin Column with the Collection Tube cap and incubate the assembly for 60 min at room temperature to allow thorough wetting of the whole blood sample on the collection disc. The 60 min incubation step is critical for obtaining a high yield of whole blood components from the collection disc.
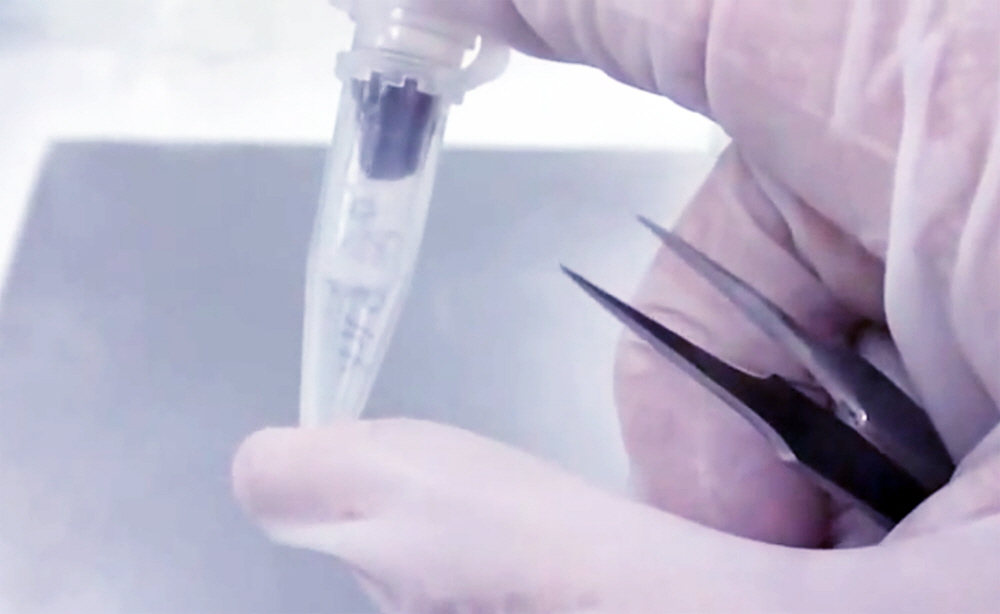
Figure 4. Arrayit Whole Blood Card Spin Column containing a 12.5 mm collection disc wetted with 60 µl of 1X Elution Buffer placed in a 1.5 ml Collection Tube containing 60 µl of 1X Reaction Buffer. The Spin Column-Collection Tube assembly should be incubated for 60 min at room temperature to allow re-hydration of the whole blood components for efficient elution.
8. Spin 3 min to collect whole blood sample and mix by vortexing. Place the Spin Column-Collection Tube assembly in a high-speed microfuge and centrifuge at 14,000 x g for 3 min to elute the whole blood components off the collection disc and into the 60 µl of 1X Reaction Buffer at the bottom of the 1.5 ml Collection Tube. A properly eluted whole blood sample will appear as a dark red 120 µl sample as shown in Figure 5. The 120 µl whole blood sample should be mixed using moderate vortexing for 5 sec to ensure proper mixing of the elution buffer, reaction buffer and whole blood components.
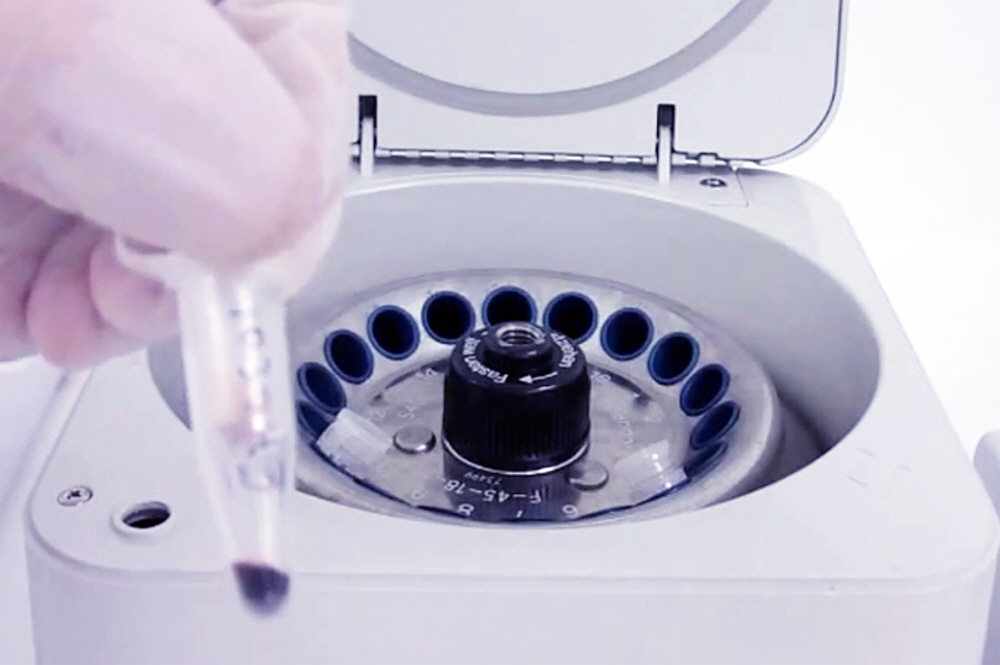
Figure 5. Arrayit Whole Blood Card Spin Column-Collection Tube assembly containing an eluted 120 µl whole blood sample in the bottom of the 1.5 ml Collection Tube. Elution is performed using centrifugation at 14,000 x g for 3 min.
9. React whole blood sample with protein microarray for 60 min. Place a printed and blocked protein microarray substrate slide (1 x 25 x 76 mm) into a hybridization cassette (e.g. Cat. AHC4X24) and react with 100 µl of whole blood sample for 60 min at 37°C with 500 rpm mixing. Array Plate Hybridization Stations (Cat. MMHS2 and MMHS4) are strongly recommended for the protein microarray reaction and staining steps. After the 60 min reaction step, wash the microarrays 6 times in rapid succession using 250 µl Protein Microarray Wash Buffer (Cat. PMWB) dispensed using a PipetteIt™ 8-Channel High-Speed Dispenser (Cat. PPIT). Invert and tap the hybridization cassette firmly on the lab bench to remove residual wash buffer.
10. Process and scan the protein microarray to obtain test results. For protein microarrays involving antibody binding, stain the protein microarray with a fluorescent secondary reagent (e.g. Cyanine 5-anti-human IgG) at a 1:5,000 dilution for 60 min at 37°C with 500 rpm mixing using an Array Plate Hybridization Station as in Step 9. After staining, wash the microarray 6 times in rapid succession using 250 µl Protein Microarray Wash Buffer (Cat. PMWB) dispensed using a PipetteIt™ 8-Channel High-Speed Dispenser. Remove the microarray slide from the hybridization cassette, dunk in distilled water for 3 sec, dry for 5 sec by centrifugation in a Microarray High-Speed Centrifuge (Cat. MHC) and scan for fluorescence emission in the cyanine 5 channel using an Arrayit InnoScan® Microarray Scanner. Quantify the scanned image using Mapix® software to obtain the test results. A properly performed protein microarray experiment using a whole human blood sample will generate the data shown in Figure 6.

Figure 6. Arrayit protein microarray data using a whole blood sample collected and eluted from an Arrayit Whole Blood Card (Cat. DBSC). The microarray was scanned using an Arrayit InnoScan® 710AL Microarray Scanner (Cat. 710AL) in the 635 nm cyanine 5 channel at 5 µm scan resolution, laser high and the PMT set at 100%.
Recommended Products and Services
NanoPrint™ 2 Microarray Printers
Arrayit InnoScan® Microarray Scanners
Arrayit Plate Hybridization Stations
Microarray High-Speed Centrifuge
PipetteIt™ 8-Channel Liquid Dispenser
Multi-Well Hybridization Cassettes
Microarray Cleanroom Wipes
Protein Microarray Buffers
SuperEpoxy 3 Microarray Substrate Slides
Protein Microarray Manufacturing Services

